AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Fda pauses johnson and johnson vaccine3/23/2023 
Vaccine safety monitoring will continue and any new information about TTS will be brought to ACIP as needed. 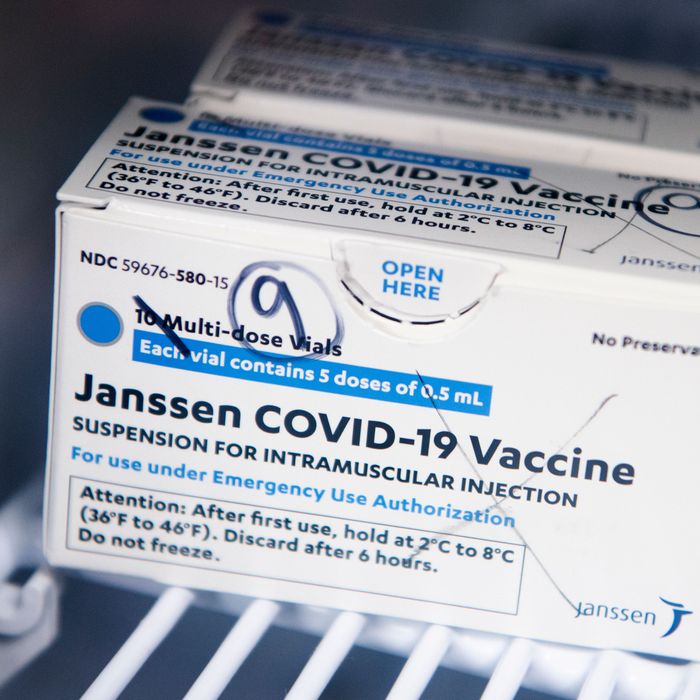
ACIP is committed to be vigilant and responsive to additional information that could impact the risk benefit analysis of any of these vaccines. Health care providers administering the vaccine and vaccine recipients or caregivers should review the Janssen COVID-19 Vaccine Fact Sheet for Healthcare Providers Administering Vaccine (Vaccination Providers) and Fact Sheet for Recipients and Caregivers, which have been revised to include information about the risk of this syndrome, which has occurred in a very small number of people who have received the Janssen COVID-19 Vaccine.ĬDC’s independent Advisory Committee on Immunization Practices met today to discuss the latest data on TTS, hearing from the vaccine manufacturer Janssen and the COVID-19 Vaccine Safety Technical (VaST) Subgroup, as well as a risk benefit analysis.At this time, the available data suggest that the chance of TTS occurring is very low, but the FDA and CDC will remain vigilant in continuing to investigate this risk.The FDA has determined that the available data show that the vaccine’s known and potential benefits outweigh its known and potential risks in individuals 18 years of age and older.The FDA and CDC have confidence that this vaccine is safe and effective in preventing COVID-19.Use of the Janssen COVID-19 Vaccine should be resumed in the United States.The two agencies have determined the following: The teams at FDA and CDC also conducted extensive outreach to providers and clinicians to ensure they were made aware of the potential for these adverse events and could properly manage and recognize these events due to the unique treatment required for these blood clots and low platelets, also known as thrombosis-thrombocytopenia syndrome (TTS). During the pause, medical and scientific teams at the FDA and CDC examined available data to assess the risk of thrombosis involving the cerebral venous sinuses, or CVST (large blood vessels in the brain), and other sites in the body (including but not limited to the large blood vessels of the abdomen and the veins of the legs) along with thrombocytopenia, or low blood platelet counts. The pause was recommended after reports of six cases of a rare and severe type of blood clot in individuals following administration of the Janssen COVID-19 Vaccine. should be lifted and use of the vaccine should resume. Centers for Disease Control and Prevention have determined that the recommended pause regarding the use of the Johnson & Johnson (Janssen) COVID-19 Vaccine in the U.S. Food and Drug Administration and the U.S. We are grateful to the researchers and scientists working to ensure that all medications or vaccines, including the COVID-19 vaccines, have the highest safety standards.Following a thorough safety review, including two meetings of the CDC’s Advisory Committee on Immunization Practices, the U.S. People who don't have a medical provider can call 2-1-1 to connect with a healthcare provider. People who received the vaccine in the last 3 weeks should look for any symptoms of these unusual clots, including severe headaches, abdominal or leg pain, and shortness of breath, and contact their medical provider if symptoms develop. These reactions are extremely rare, as nearly 7,000,000 people have received the Johnson & Johnson vaccine in the United States to date. Vaccine Providers in Los Angeles County will contact patients about rescheduling or providing alternative vaccine brands. This pause will last until the FDA and CDC complete their review, which is expected to take several days. 
LADPH Press Release - Out of an abundance of caution, Los Angeles County is following the recommendation of the Food and Drug Administration (FDA) and the Centers for Disease Control (CDC) to pause the use of the Johnson & Johnson vaccine after reports that 6 women between the ages of 18 and 48 developed unusual types of blood clots 6 to 13 days after receiving the vaccine. The CDC and FDA have released a communication on the Johnson & Johnson vaccine status, details below: 
If you have any concerns or questions on this alternate, please call us at (818) 963-5690. IMPORTANT NOTE: San Fernando Community Health Centers are currently administering the Moderna Vaccine to all our already scheduled appointments.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
